Introduction
Myelodysplastic syndromes (MDS) are haematological diseases characterised by clonal proliferation due to genetic and epigenetic alterations within haematopoietic stem and progenitor cells. Thus far, the prediction of a patient's overall survival (OS) and event-free survival (EFS) has been dependent predominantly on the degree of peripheral blood cytopenias, bone marrow blast percentage, cytogenetics, and genetic features. However, this may overlook other biological phenotypes, including, for example, the expression of transposable elements (TEs), ageing, and immunology profile. In this study, we present a comprehensive analysis of three data modalities (clinical, genotypic, and transcriptomic) and eight different views derived from these modalities to identify the hidden factors that significantly impact MDS prognosis.
Methods
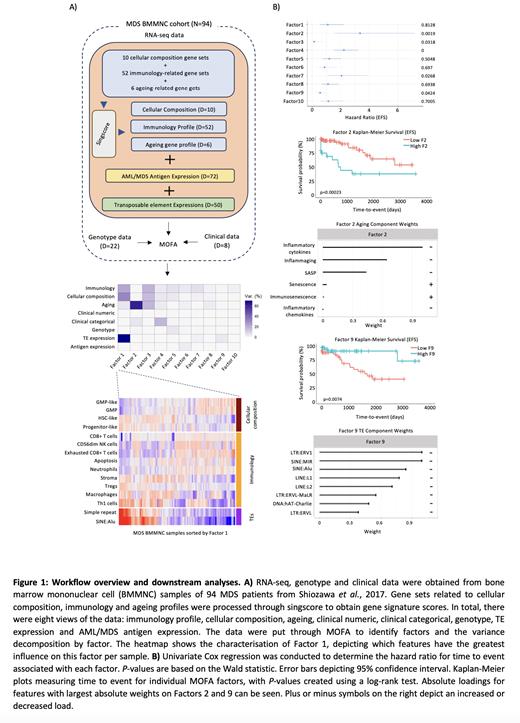
Three (immunology, ageing, and cellular composition) out of eight views of the data were derived from RNA-seq by applying singscore, a gene set scoring tool developed by Foroutan et al., 2018, on RNA-seq gene expression from bone marrow samples of 94 MDS patients obtained from Shiozawa et al., 2017 (Fig 1.A). Each of these three views was carried forward in the workflow by a number of gene sets. The other five views were clinical numeric data, clinical categorical data, TE expression, genotype data, and known AML/MDS antigens (obtained from TANTIGEN 2.0), expressed in at least 10% of MDS samples in the study. We then employed Multi-Omics Factor Analysis (MOFA), a computational framework developed by Argelaguet et al., 2018, to uncover hidden phenotypic states within a multi-omic dataset. Through MOFA, we generated factors representing specific biological data modalities, establishing connections between features across these different categories. The total variance ( R2) that can be attributed to specific factors is also exhibited by MOFA.
Results
Factors were ranked based on their explained variance. We present the 10 factors with a threshold of variance explained of 2% from the 15 default factors generated by MOFA (Fig 1.A). Factor 1 was one of the factors that linked the most categories of data: immunology, cellular composition, ageing, and TEs. Most notably, Factor 1 was aligned with features of less differentiated cells (more HSC-like and less GMP-like). We observed that samples represented by high Factor 1 scores also had a higher expression of SINE:Alu elements. (Fig 1.A). Moreover, Factor 1 aligns the following immunology features: increased T-helper 1 (Th1) cells, macrophages, and T regulatory cells. The relationship between Th1 and SINE elements is particularly interesting, given the role of Th1 cell activation in host defence against infection that may be modulated by SINE activation. Factor 2 comprised of ageing and clinical categorical data, which showed that less inflammaging may lead to a poorer EFS ( P=0.00023) (Fig 1.B). Factor 4 linked immunology, cellular decomposition, ageing and clinical categorical data. The following features mainly influenced Factor 4: increased HSC-like cells, decreased CD8+ T cells, decreased immunosenescence and increased inflammatory chemokines. Using Kaplan-Meier plots to predict OS, we observed that patients with more HSC-like cells, fewer leukocytes and T cells, and less immunosenescence and inflammaging were linked to poorer EFS ( P=0.0001) and OS ( P=0.00017, data not shown). Furthermore, Factor 9 comprised solely of the TEs category and exhibited a correlation between increased TE expression and poorer EFS ( P=0.0074) and OS ( P=0.0065, data not shown) (Fig 1.B).
Conclusions
Our analysis using MOFA on a diverse set of biological data modalities uncovered hidden factors in MDS, providing insights into the complex interplay between various biological layers. Notably, we identified TE expression as a risk factor and inflammaging as a protective factor in MDS. HSC-like cells and specific immunological profiles were also associated with significantly poor OS and EFS. This study contributes to a deeper understanding of MDS pathogenesis and identifies potential prognostic markers for this disease. It also elucidates the importance of considering the relationships between different pathways, markers, and mutations in predicting patient outcomes, highlighting the efficacy of a comprehensive approach that goes beyond all the scoring systems described thus far for MDS.
Disclosures
No relevant conflicts of interest to declare.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal